| -250% CO2 emission until 350 ppm are reached again
|
| 
|
|
|
Less CO2 emission is much too little, even zero emission is insufficient. Only a planet renovation with large-scale CO2 filtering and splitting from the atmosphere will help.
|
| 390 PWh/year electricity for CO2 from the atmosphere
|
| 
|
|
|
Reduce the CO2 content with Power to Carbon, generate fuels with Power to Liquid and use CO2 for indoor plant cultivation to replace large-scale agriculture.
|
| Carbon fiber becomes standard material for construction
|
| 
|
|
|
If we filter the 33.1 Gt CO2 emission of 2019 from the atmosphere and split it into C and O, we get 9 billion tons of carbon. What to do with it?
|
| IPCC Report 2021: “Net Zero Emissions” Fairy Tale and its Devastating Consequences
|
| 
|
|
|
Constantly contradicting itself, the IPCC 2021 report adheres to the completely inadequate “net zero emissions” target. This can only be thinking prohibitions.
|
| Green dogmatism and the destruction of the German photovoltaic industry
|
| 
|
|
|
How the dogma “the demand for electricity will decrease” led to extremely wrong targets, which led to the destruction of the German photovoltaic industry in 2013.
|
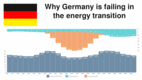
| Why Germany is failing in the energy transition
|
| 
|
|
|
In the first decade of the new millennium, Germany was the great role model in the energy transition, why, on the other hand, the current EEG policy leads to disaster.
|
| Sri Lanka crisis 2022 example of oil exit failures
|
| 
|
|
|
Hit hard by the breakdown in tourism caused by COVID-19, the higher price of oil comes as the next blow. The serious failures of the industrialized countries.
|
| Destructive cult prevents functional energy transition
|
| 
|
|
|
A horror novel about a world without renewable energy and recycling delayed a workable energy transition and thus effective climate protection by decades.
|
| Functional energy transition vs. German energy transition
|
| 
|
|
|
We have to denounce very hard the grotesque German energy transition in order to turn all the enemies of this grotesque into fans of a functional energy transition.
|
| Cost optimization — the key to energy transition and climate protection
|
| 
|
|
|
Paper written for the CORP.at conference March 22 to 25, 2026, in Vienna. My 2025 participation was a last-minute action, but now I have much time to prepare the paper.
|









